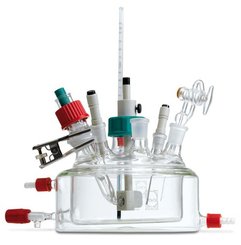
Reference Electrode built-in Pseudo Reference Electrode and RHE
|
A reference electrode is an electrode whose potential is constant and can be related to a fundamental thermodynamic scale, e.g. the standard hydrogen electrode. Reference electrodes are widely used in electrochemistry as a standard against which to measure the potential of other electrodes
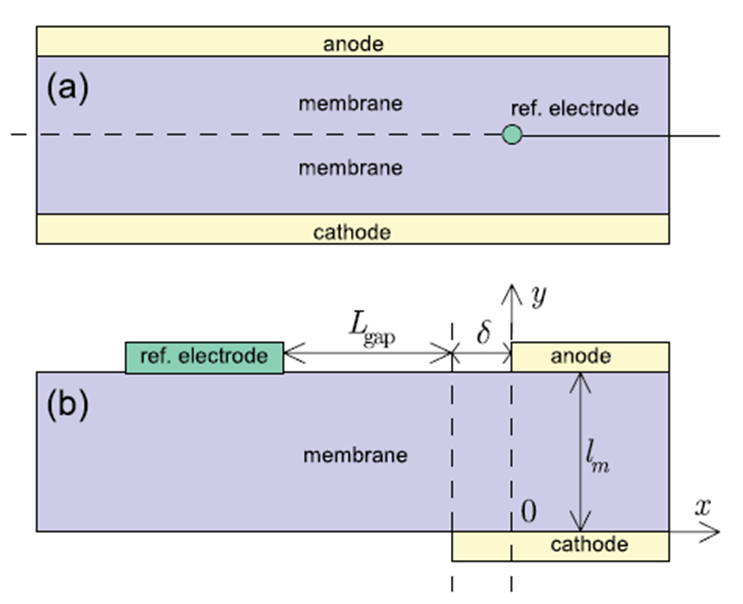
A Three-Electrode configuration for an electrochemical cell is most common for typical electrochemical applications. A third electrode (the reference electrode) is used to determine the potential across the electrochemical interface accurately. Since the absolute potential of a single electrode cannot be measured, all potential measurements, in electrochemical systems are performed with respect to a reference electrode. The state of the art in reference electrodes for solid polymer fuel cells Three common reference electrodes used in solid polymer fuel cell systems are pseudo-reference electrode, reversible hydrogen electrode and dynamic hydrogen electrode. Uncertainty in the reliability of reference electrode measurements in solid polymer fuel cell is mainly associated with the positioning of the reference electrode with respect to the fuel cell electrodes, the stability of the reference electrode and perturbation of fuel cell operation by the reference electrode. Reference electrodes may be positioned either internal or external to the fuel cell. Internal reference electrodes are typically constructed by sandwiching the electrode between two membranes, which may perturb fuel cell operation. External reference electrodes are usually connected to the fuel cell membrane by a salt bridge. The installation of a reference electrode inside a fuel cell unit as a diagnostic tool along with electrochemical impedance spectroscopy (EIS) can potentially detect subtle changes in fuel cell structure and provide useful information about rate-limiting processes |
“Positioning of a Reference Electrode in a PEM Fuel Cell”, A. A. Kulikovsky and P. Berg, Journal of The Electrochemical Society, 162(2015)F843.
|
Fuel cells show potential - Pseudo and RHE
Ecscell introduces fuel cell with innovative internal reference electrode that is used to measure changes in electrode potential inside a working polymer electrolyte membrane (PEM) fuel cell.
The internal reference include Pseudo-Reference Electrode and the Reversible Hydrogen Electrode (RHE). This RHE electrode consists of a platinum electrode dipped in a solution of 1 M acid (usually HCl) with hydrogen gas at a partial pressure of 1 atm bubbled along its surface.
Ecscell introduces fuel cell with innovative internal reference electrode that is used to measure changes in electrode potential inside a working polymer electrolyte membrane (PEM) fuel cell.
The internal reference include Pseudo-Reference Electrode and the Reversible Hydrogen Electrode (RHE). This RHE electrode consists of a platinum electrode dipped in a solution of 1 M acid (usually HCl) with hydrogen gas at a partial pressure of 1 atm bubbled along its surface.