CO2 Reduction Reaction (CO2RR) is an electrochemical reduction of CO2 that convert CO2 into value-added products, including CO, formic acid, methanol, ethylene, alkanes, and alcohols. Substantial efforts have been invested to determine the special catalytic properties including heterogeneous and molecular electrocatalysts to elucidate the mechanism through which hydrocarbons are formed.
- from H-cell to Flow-cell reactors
- understanding of the catalytic mechanism
- design of efficient electrocatalysts to control the reaction pathway
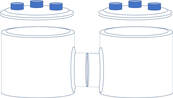
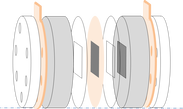
H-cell is widely used for screening electrocatalysts.
- Study reaction mechanisms, material structures, surface sites, and local pH effect
- Conduct cyclic voltammetry (CV) for the investigation of electrocatalytic CO2 Reduction
- Furnish photocatalytic CO2 reduction
Flow-cell reactor maximizes CO2 reduction formation and current density.
- Introduce CO2 in the gas phase or dissolved in aqueous media
- Gas-phase CO2 enhances mass transfer vs. liquid-dissolved CO2
- Apply aqueous electrolytes in the CO2RR include alkali, ammonium, sulfate and salts
- Build advanced vaper-feed reactors for gas phase CO2 electrolyzer
CO2 reduction - Cation, Anion polymer electrolyte and liquid electrolyte
For the electrochemical CO2RR from gas phase with cation/anion exchange membrane, relative humidity is one of the critical elements for product conversion, selectivity and process efficiency.
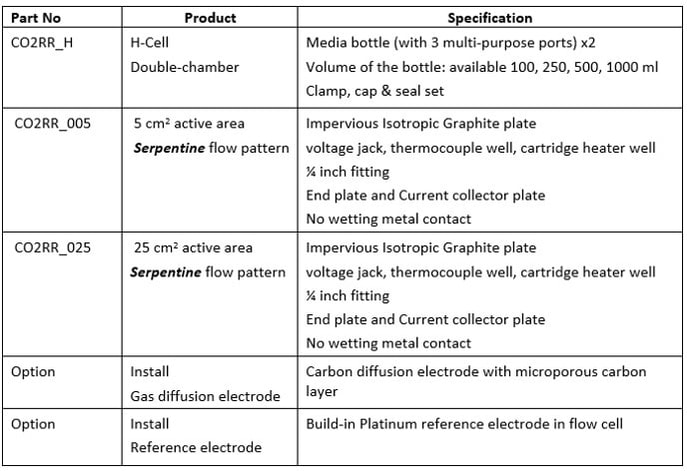
CO2 Reduction Cell
Designed to allow the build of multi-cell stack and built-in cooling plate
Features
|
Applications
|
Accessories
|
Part Number
|
Product
|
Specification
|
|
Gas diffusion layer
|
Carbon paper and Titanium felt
|
|
Polymer Electrolyte Membrane
|
Cation and anion membrane
|
|
Gasket
|
Precision flat gasket
|
|
872-216-7641
|
Ecscell.com
|